Welcome to Rejuvenation Science®!
1-888-737-3588 TOLL FREE
Welcome to Rejuvenation Science®!
Ten hypercholesterolemic subjects were recruited for the study. The group consisted of men and post-menopausal women with serum total cholesterol > 5.9 mmol/L, LDL cholesterol 4.0 mmol/L and serum triacylglycerols < 3.5 mmol/L (>230 mg/dL, >155 mg/dL and <307 mg/dL, respectively). To be included, participants also had to i) be free of thyroid disorders, kidney disorders and diabetes, ii) have alcohol intake not greater than two drinks per day, ii) if females, not taking hormone replacement. Subjects taking cholesterol-lowering medications were asked to discontinue the treatment 6 weeks before the study. All subjects were advised to: 1) continue with the same diet and dietary habits that they had prior to the study and, 2) avoid taking supplements such as vitamins, minerals etc. six weeks before and during the study.
Participants were asked to take SYTRINOL™ supplement consisting of PMF (tangeretin, sinensetin, nobiletin) and tocotrienols each day. To determine whether this treatment improved parameters associated with high risk of heart disease, fasting blood samples were drawn at the onset of the study and at the end of the 4-week trial. They were tested for plasma total and lipoprotein cholesterol, plasma apolipoproteins B (associated with LDL) and A1 (associated with HDL), total triacylglycerols. The protocol was approved by the Human Ethics Committee of the University of Western Ontario and informed consent was obtained from each subject.
Cholesterol and triacylglycerols were measured with enzymatic timed-endpoint methods by using CHOL Reagent or Triacylglycerol GPO reagent. Plasma concentrations of apo B and apo A1 were analyzed immunoephelometrically with a BNII System.
Subjects were instructed to maintain their caloric intake during the study. This was ensured by measurement of Body Mass Index (BMI) before and after treatment.
Changes from baseline after 4 weeks were analyzed by using repeated-measures analysis of variance (ANOVA) followed by Dunnett’s t tests.
Changes in the subject’s baseline characteristics during treatment are presented in Table 1.
Table 1. Effect of Sytrinol supplement on serum and lipoprotein cholesterol levels in hypercholesterolomic human subjects
| Variable | Baseline | 4 weeks |
| Body mass index (kg/m2) | 28.8 ± 4.6 | 28.6 ± 4.5 |
| Total cholesterol mg/Dl | 266.82 ± 34.80 | 201.08 ± 27.07 |
| VLDL cholesterol mg/dL | 30.94 ± 11.6 | 34.80 ± 15.47 |
| LDL cholesterol mg/dL | 181.75 ± 30.94 | 146.95 ± 27.07 |
| HDL cholesterol mg/dL | 42.54 ± 11.60 | 42.54 ± 11.60 |
| Total triacylglycerol mg/dL | 96.68 ± 19.34 | 73.47 ± 19.34 |
Means ± SEM. Statistical analysis by ANOVA plus post test by Dunnett’s method.

To evaluate the cardio protective potential of SYTRINOL™ in subjects with moderate hypercholesterolemia
10 subjects (7 men and 3 women) with a mean (±SD) age of 52 ±3 years and average body weight of 80.8 ± 10.9 kg were recruited from two clinics. All participants had elevated initial plasma total and LDL cholesterol concentrations (6.61 ± 0.93 and 5.10 ± 0.88 mmol/L, respectively). To be eligible for participation, individuals had to i) have initial fasting plasma triacylglycerol concentration less than 3.5 mmol/L, ii) be free of thyroid disorders, kidney disease and diabetes, iii) have an alcohol intake of two or less drinks per day. The participants were not on cholesterol-lowering medications for at least 6 weeks prior to study and during the study. They were also asked to avoid taking supplements (e.g. vitamins, minerals, herbal products) during the study. The experimental protocol was approved by the Human Ethics Committee of the University of Western Ontario.
Subjects eligible for the study were asked to take SYTRINOL™ capsules for 4 weeks. Daily dose was 300 mg (2 capsules, each containing 30 mg tocotrienols, 270 mg of polymethoxyflavones taken as 1 in the morning, 1 at night). Blood samples were taken from the forearm vein before the start (baseline), and at the end of 4 wk treatment period. Plasma lipids profiles and other metabolic parameters were analyzed using standard methods. Blood pressure was recorded in the sitting position, using a conventional mercury manometer, by calculating a mean of two readings.
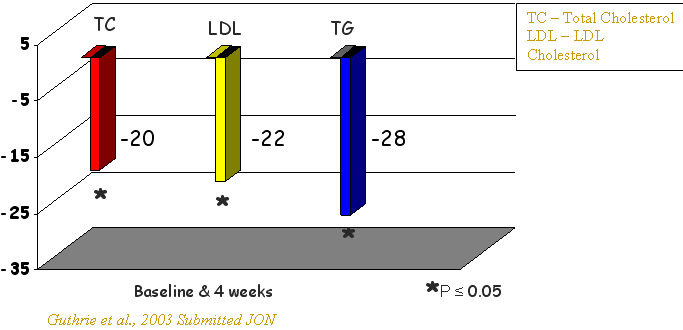
The results of the study are summarized in Table 1. Our data presented in Table 1 demonstrated that in hypercholesterolemic subjects, dietary supplementation with 300 mg SYTRINOL™ /day for 4 weeks resulted in a number of beneficial effects. Treatment with SYTRINOL was associated with significant reduction in plasma total cholesterol (19.7%), LDL cholesterol (22.01%) and apo B (20.9%) (protein component of VLDL and LDL). A significant 28.4% reduction of serum triacylglycerols and a significant 5% increase of apo A1, a structural protein of HDL was observed.
Table 1. Blood pressure, BMI and blood lipid concentrations at baseline and after treatment period
| Variable | Week 0 | Week 4 |
| Systolic blood pressure, mmHg | 123.0 ± 22.4 | 116.0 ± 12.7 |
| Diastolic blood pressure, mmHg | 79.0 ± 13.0 | 76.0 ± 9.2 |
| Body weight, kg | 80.8 ± 10.9 | 81.1 ± 11.7 |
| Body Mass Index (BMI), kg/m2 | 27.4 ± 1.8 | 27.5 ± 1.9 |
| Total cholesterol, mmol/L | 255.61 ± 35.96 | 205.34 ± 18.95* |
| Triacylglycerols, mmol/L | 77.34 ± 29.78 | 55.41 ± 13.64* |
| HDL cholesterol, mmol/L | 42.92 ± 9.67 | 44.08 ± 8.51 |
| LDL cholesterol, mmol/L | 197.60 ± 34.03 | 154.29 ± 16.63* |
| Apo A1, g/L | 49.50 ± 5.8 | 51.82 ± 6.19* |
| Apo B, g/L | 51.82 ± 6.96 | 40.99 ± 4.25* |
* - significantly different between baseline and week 4.
Sytrinol™ should be taken as part of a diet low in saturated fat and cholesterol.
Please consult with your Doctor or Pharmacist regarding your personal condition, drug and supplement use. Do not discontinue the use of a prescription drug without the advice of your doctor.